This is an old revision of the document!
Example: Attempting to Charge Insulators by Induction
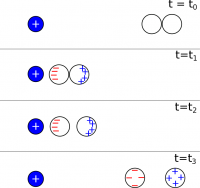
In the notes on Charges and Matter, we saw how to charge a pair of conductors using induction. The relevant figure is copied here.
Facts
- The Avogadro constant is $N_A = 6.022 \cdot 10^{23} \text{ mol}^{-1}$
- Note: When we write the unit as $\text{ mol}^{-1}$, we mean particles per mole. We could also write this unit as $mol^{-1}=\frac{1}{mol}$.
- All electrons have the same charge, which is $e = -1.602\cdot10^{-19} \text{ C}$.
Lacking
- Total Charge
Approximations & Assumptions
- None, we have all the information we need.
Representations
- The total number of particles $N$ can be found from the number of moles $m$ using the Avogadro constant: $N = m \cdot N_A$.
- The total charge $Q$ can be written as the number of particles $N$ times the charge of each particle ($e$, for electrons): $Q=N\cdot e$.
Solution
yeas